Overview
Sacral neuromodulation (InterStim therapy) involves the implantation of a small device that delivers mild electrical pulses to the sacral nerves, which control bladder and bowel function. It is used to treat chronic urinary retention, refractory overactive bladder symptoms, and fecal incontinence.
Two-Stage Approach
Stage 1: Test Phase
A thin, flexible lead (wire) is placed near the sacral nerve under fluoroscopic guidance. The lead is connected to an external stimulator worn on the belt. During a trial period (typically 1-2 weeks), you can assess whether your symptoms improve.
Stage 2: Permanent Implant
If symptoms improve by at least 50% during the trial, a small battery-powered pulse generator (similar to a pacemaker) is implanted under the skin in the upper buttock area and connected to the lead.
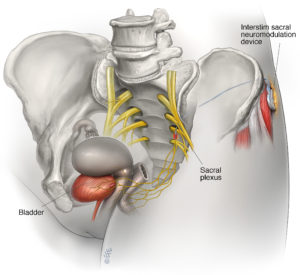
Indications
- Overactive bladder unresponsive to conservative and medical treatments
- Urgency urinary incontinence
- Non-obstructive urinary retention
- Fecal incontinence unresponsive to other treatments
Effectiveness
- Approximately 70-80% of patients who proceed to permanent implant achieve significant symptom improvement
- The device can be adjusted over time via an external programmer
- Battery life: typically 5-10 years, after which the generator is replaced
Considerations
- MRI compatibility varies by device generation — discuss with your doctor
- The device requires periodic follow-up for adjustment and monitoring
- This is reserved for patients with severe, persistent symptoms who have not responded to other treatments
Risks and Success Rates
Sacral neuromodulation achieves cure or significant improvement in up to 80% of patients with overactive bladder or urinary retention. However, approximately one-third of patients may require further surgery due to device-related issues. Potential risks include:
- Pain — may radiate to the back, buttock, or thigh
- Skin irritation at the implant site
- Infection — at the device or lead site
- Lead migration — the electrode may shift from its original position
- Uncomfortable stimulation — stimulation settings may need adjustment
- Device malfunction — technical failures may require revision surgery
- Undesirable changes in bowel or voiding function
Illustrations courtesy of the International Urogynecological Association (IUGA) Patient Information Leaflets.